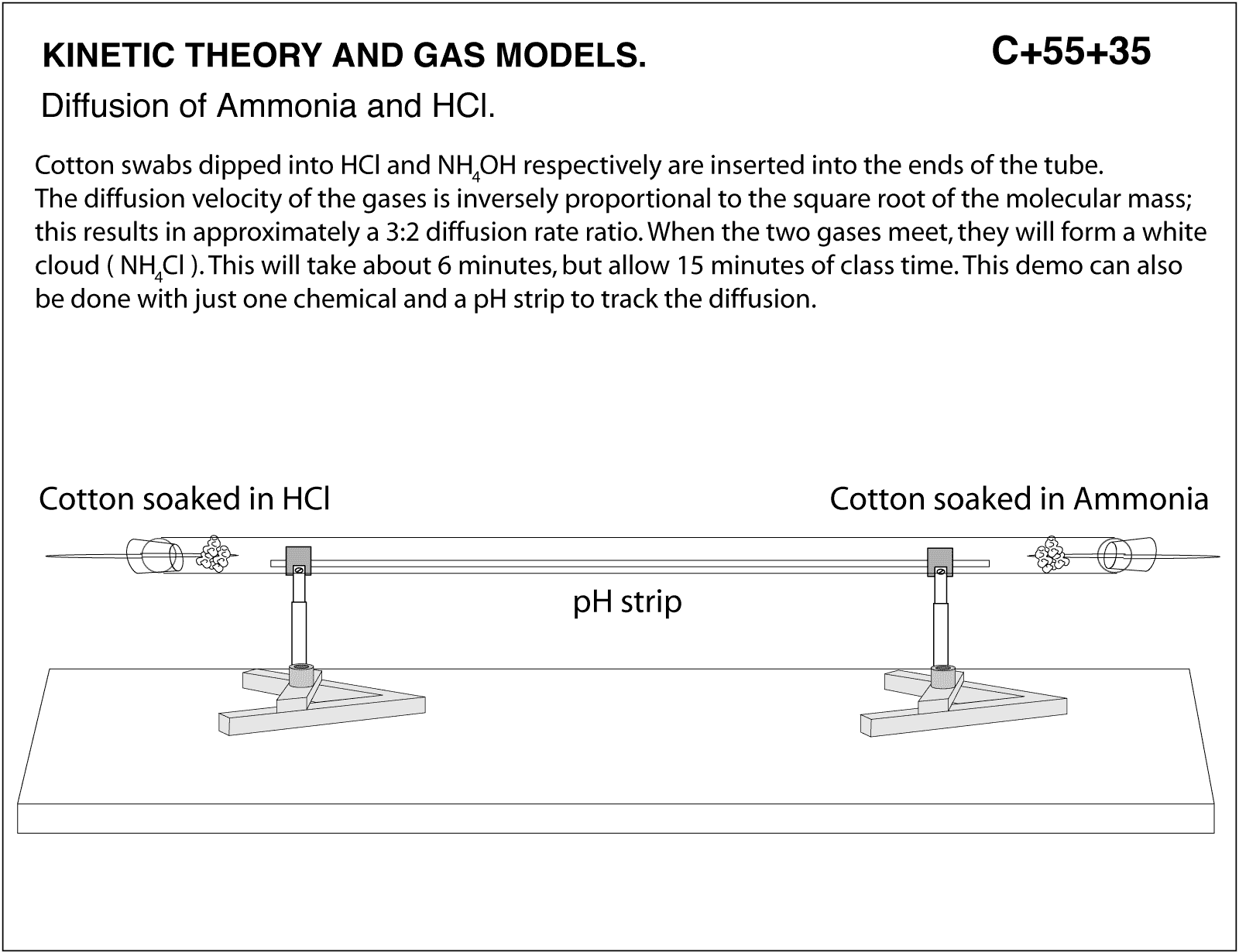
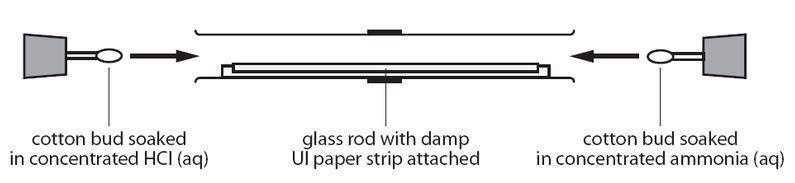
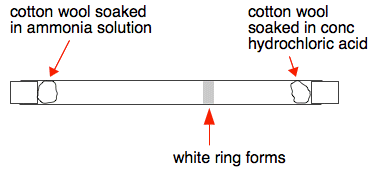
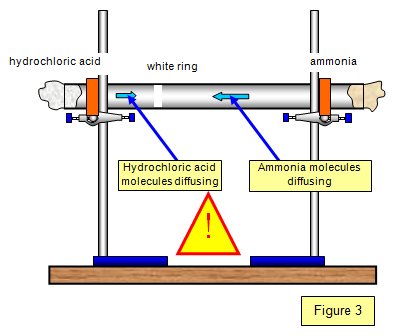
In a gaseous reaction between ammonia and hydrochloric acid (HCl), a white precipitate of ammonium chloride is produced according to the following reaction. NH3(g) + HCl (g) rightarrow NH4Cl(s) Two cotton plugs

A bottle of perfume is opened in the room why can we smell it after a while This is called diffusion Diffusion is when particles move from a high. - ppt download
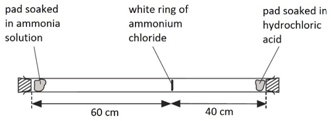
1:03 understand how the results of experiments involving the dilution of coloured solutions and diffusion of gases can be explained - TutorMyself Chemistry
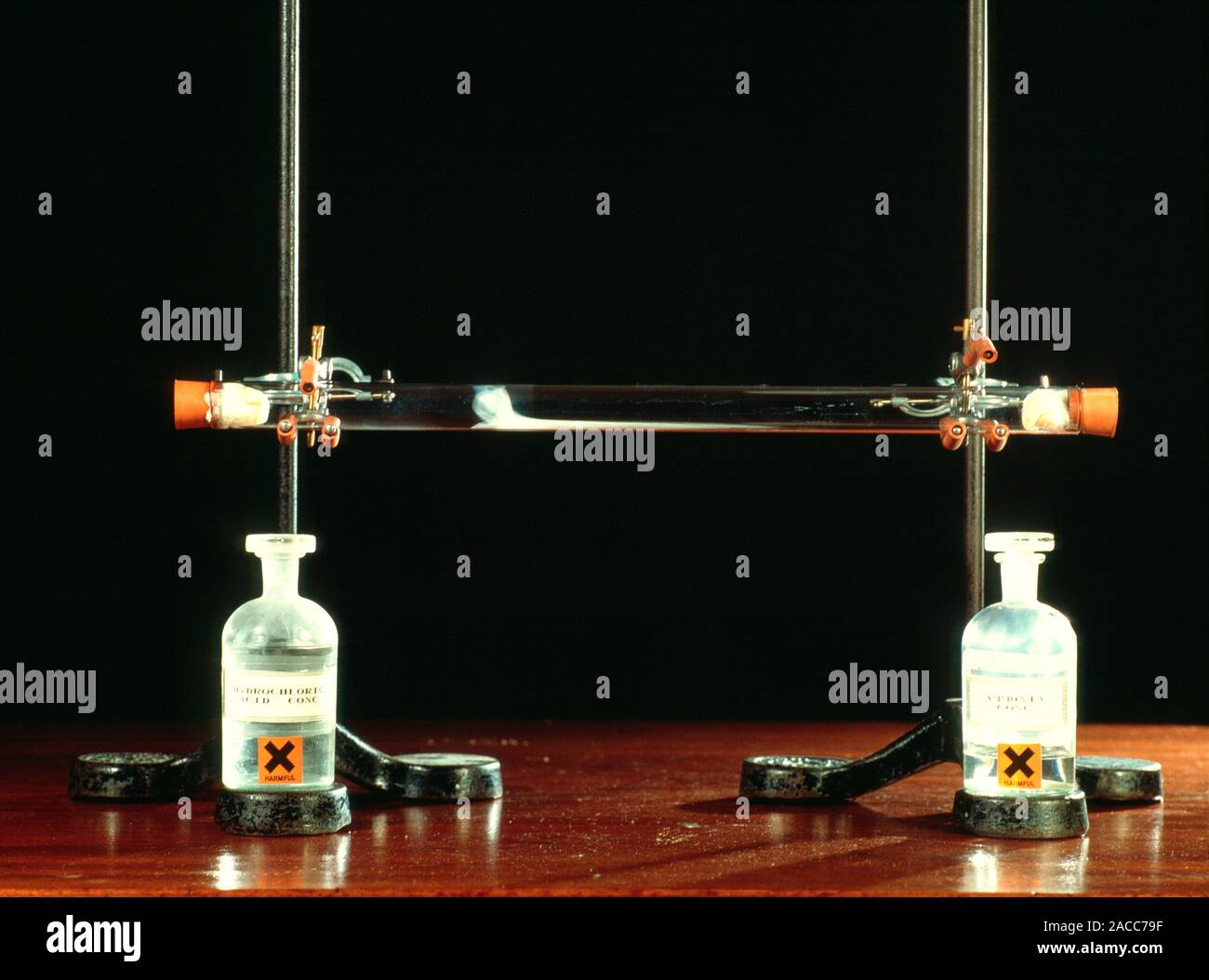
Ammonium chloride formation. Cotton wool balls have been soaked in hydrochloric acid (HCl, left) and ammonia (NH3, right). They have been placed at ei Stock Photo - Alamy
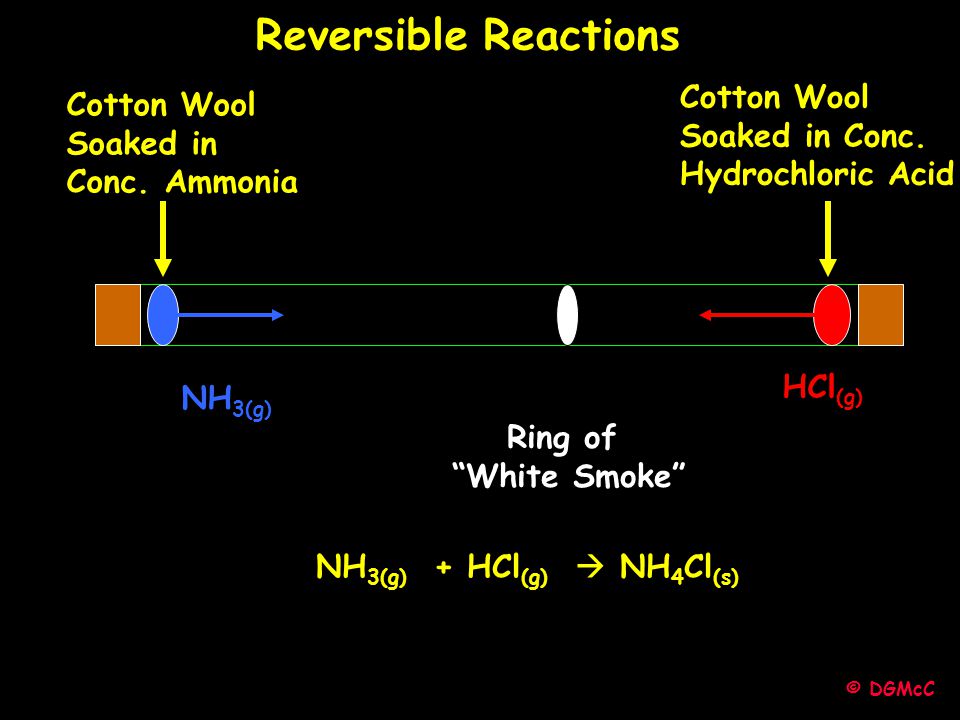
DGMcC Magee Lecture. © DGMcC Cotton Wool Soaked in Conc. Ammonia Cotton Wool Soaked in Conc. Hydrochloric Acid NH 3(g) HCl (g) Ring of “White Smoke” - ppt download

Look at the glass tube below Suppose it was warmed a little in an oven before the experiment Do you think that would change the result If so how The particles in













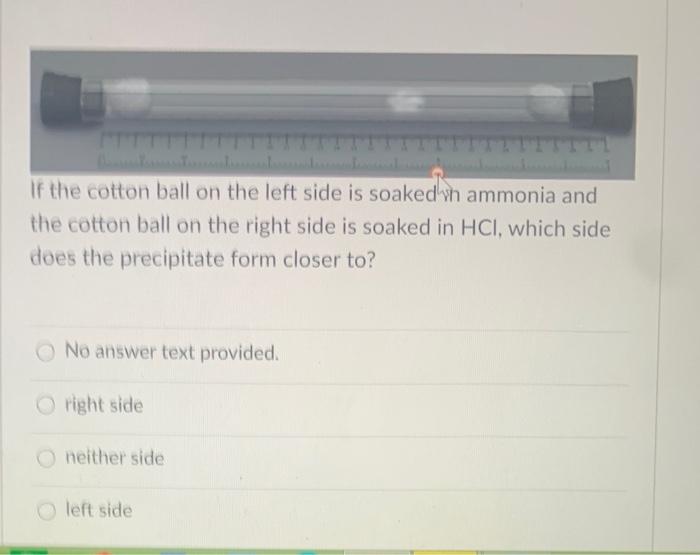
![Solved] The apparatus shown below was set up. Giv | SolutionInn Solved] The apparatus shown below was set up. Giv | SolutionInn](https://s3.amazonaws.com/si.question.images/images/question_images/1629/7/9/1/5716124a5537b1dc1629791571021.jpg)